Bacteriostatic Water. Pharmaceutical Grade. Bulk Supply.
USP-grade bacteriostatic water for research facilities, compounding labs, and wholesale distributors. Batch-traced, independently tested, shipped from an FDA-registered facility.
500+
Minimum Order
USP 797
Compliant
COA
Batch-Traced
FDA
Registered Facility
Wholesale Product Lines
All vials manufactured in an FDA-registered, cGMP-compliant facility. Each lot ships with a Certificate of Analysis.
BAC Water 10ml
Compact vials for research applications requiring smaller volumes. Sterile, multi-dose format with 0.9% benzyl alcohol preservative.
Volume Pricing
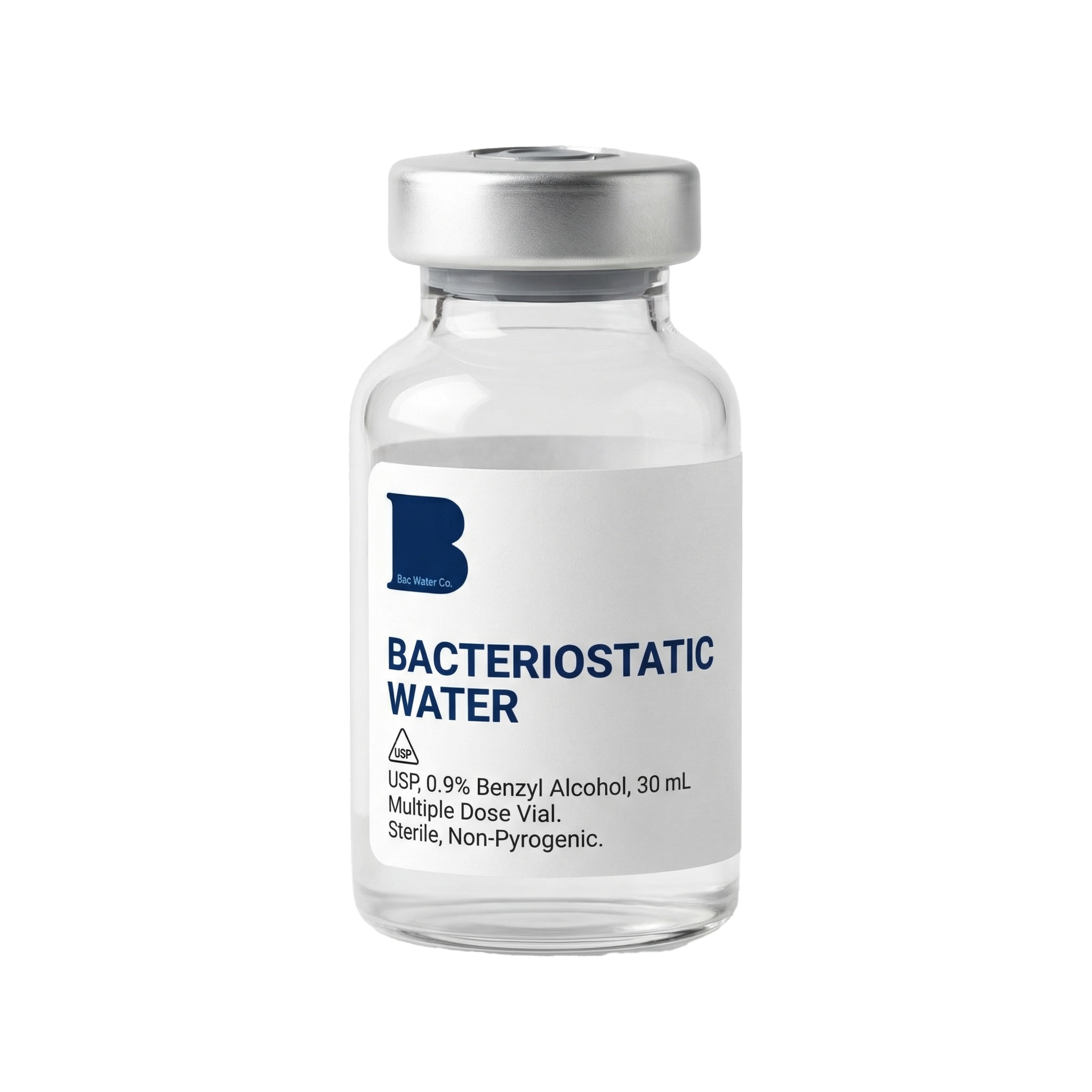
BAC Water 30ml
Our standard research-grade vial. Most popular among wholesale partners for lab supply and distribution channels.
Volume Pricing
BAC Water 50ml
Larger format vials for high-volume reconstitution workflows. Ideal for facilities with elevated throughput requirements.
Volume Pricing
Pharmaceutical-Grade Standards
Every vial meets the quality benchmarks your facility expects. Full documentation, independent verification, complete traceability.
Certificate of Analysis
Every production lot ships with a full COA documenting sterility, endotoxin levels, pH, and preservative concentration. Available on request before order.
USP 797 Compliance
Manufacturing processes adhere to United States Pharmacopeia Chapter 797 standards for sterile compounding and preparation.
FDA-Registered Facility
Produced in a facility registered with the U.S. Food and Drug Administration, following current Good Manufacturing Practice (cGMP) guidelines.
Independent Testing
Third-party laboratory verification for every batch. Sterility, particulate, and endotoxin testing performed by accredited independent labs.
Lot Traceability
Full chain-of-custody tracking from raw materials through finished product. Every vial is traceable to its production lot, date, and facility.
Consistent Supply
Dedicated production capacity with standing inventory for wholesale partners. No backorders, no allocation limits, reliable recurring shipments.
Built for labs that
can't afford inconsistency.
Bac Water Co. was founded to solve a supply chain problem: research facilities and wholesale distributors need pharmaceutical-grade bacteriostatic water with reliable availability, verifiable quality, and pricing that scales.
We manufacture exclusively in FDA-registered, cGMP-compliant facilities. Every production run follows USP 797 protocols. Every lot is independently tested and ships with a full Certificate of Analysis.
Our wholesale partners range from compounding supply distributors to university research procurement departments. We understand the documentation requirements, the lead time expectations, and the zero-tolerance quality standards your work demands.
100K+
Vials shipped monthly
99.9%
Order accuracy
48hr
Standard lead time

cGMP Manufacturing
Net-30 Terms
Account Support
Private Label
Request Wholesale Pricing
Tell us about your needs and we'll prepare a custom quote. Minimum order: 500 units.
Accepting new wholesale partners